How are Diamonds Formed?
You might be intrigued to know that how are diamonds formed. Well, in that case you are at the right place.
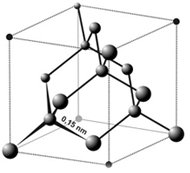
Atomic Structure of Diamond
 |
| Atomic Structure of Diamond |
Diamond is the only gem that is made of a single element, i.e. pure carbon. Carbon is denoted as ‘C’ in the field of chemistry. Diamond is a transparent crystal composed of tetrahedral-bonded carbon atoms.
How are Diamonds Formed?
This gem is formed at high temperature of 1500oC and at pressure of around 5 Giga Pascal. These extreme conditions exist approximately 100 miles beneath the earth’s surface.
It takes millions of years for a piece
of diamond to reach its current state from carbon containing minerals.
These carbon containing minerals are in abundance in earth’s Mantle
(layer of earth which lies between innermost Core and outermost Crust). You must also read about how diamonds come to earth's surface.
But unfortunately, all carbon containing minerals do not turn into diamond. Only those which are exposed to above-mentioned conditions get converted to this rare gem. Even if there is a slightest of change in temperature or pressure, final outcome doesn’t results to diamond.
Diamond passes through lots of chemical processes over the period of its long journey of formation. So, one can rightly say that, ‘Diamond is a Legend in itself’.
Of course, this can’t be said for every diamond. After all, not every diamond takes millions of years to reach its sparkling form. Now, you may ask how?
Man made diamonds or Imitation Diamonds are the answer. Increasing demand of diamond has led to the creation of Imitation Diamond. It includes all those crystals that are artificially created in laboratory.
Man-made diamond can be broadly categorized into:
- Synthetic Diamond
- Diamond Simulant
Synthetic Diamonds are the crystals that are grown in laboratory. Synthetic diamonds have more or less same physical, chemical and optical properties as natural diamonds.
 |
| Synthetic Diamonds |
These crystals are produced in laboratory using special techniques like High Pressure High Temperature (HTHP), Chemical Vapor Deposition (CVD) etc.
Synthetic diamonds are categorized into different man made diamond types according to their manufacturing process. Learn more about Synthetic Diamonds...
Diamond Simulant or “Diamante” is actually not a diamond. It is used to simulate the appearance of diamond. Unlike diamond, it is not in pure carbon form. Rather it is composed of different compounds.
The common simulants are rhinestones (high-leaded glass), Cubic Zirconia (Zirconium Oxide, ZrO2) or CZ and Moissanite (Silicon Carbide, SiC). Here rhinestones and CZ is artificially created whereas Moissanite is a natural gem.
Return from How are Diamonds Formed to Diamond Jewelry Homepage
I hope you'll not mind sharing this on Twitter, Facebook and with everyone else :)
Feel free to share if something is in your mind and want it to be covered on this site.
My Newsletter
Did you liked this article? Sign-up my FREE weekly newsletter and I'll send you more awesome new additions on this website along with latest jewelry happenings around the world, and download my Jewelry Design Album for FREE!
 |
|