Hardness of Diamond - Can diamond resist breakage?
Hardness of diamond implies its resistance to scratching.
Diamond is the hardest natural mineral found on this planet. Diamond derives its name from Greek word A’da’mas - which means 'unbreakable' - referring to its hardness. In Sanskrit it is called Vajra meaning 'thunderbolt'.
You may also like to read about how are diamonds formed and how diamond comes to earth’s surface.
What is Diamond's Hardness?
On Mohs Scale, diamond’s hardness is 10 (1 is the softest and 10 is the hardest grade), while its absolute hardness is 1600. So, in terms of absolute hardness, it is 4 times harder than Corundum - the next hardest substance from which rubies and sapphires are formed.
Reason behind Hardness is Diamond Structure
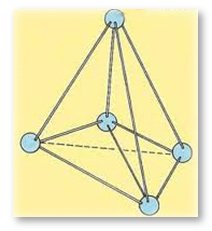
Diamond is formed of tetrahedral bonded carbon atoms with 1 carbon atom linked to 4 other carbon atoms. This bond structure forms an inflexible three dimensional lattice and it is the prime reason behind the hardness of diamond.
Diamond's Molecular Tetrahedral Structure
What is the main reason behind difference between properties of Diamond and Graphite?
This is a very common and obvious question, after all hardness of diamond and graphite are at the extreme end of spectrum, even after both being composed of carbon.
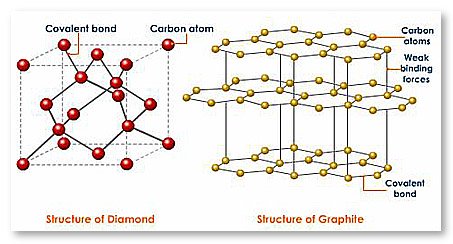
Chemically, diamond and graphite are identical (i.e. both are composed of carbon), but these two substances are quite opposite in terms of their physical properties. While, former is the hardest natural substance, latter is very soft in nature.
Reason behind graphite being soft is the way carbon atoms are bonded in it. In graphite, carbon atoms are arranged in hexagonal ring pattern in a 2-D plane (like a flat sheet). Although, bonding between different carbon atoms is strong, bonding between sheets (of hexagonal arranged carbon atoms) is relatively weak. Due to this, sheets can easily slide over each other, thereby making graphite a soft substance.
Confusion between Hardness and Toughness
"Hardness" of a substance means its resistance to scratching. So, when we say that diamond is the hardest substance, then it means that it can’t be scratched by any material except another diamond. On the other hand, "Toughness" of a substance means its ability to resist fracture or breakage when it undergoes mechanical stress or impact.
So, while diamond is the "hardest" substance, in terms of toughness it can be only moderately rated. This is because of its ability to fracture along cleavage plane. This comparison will make your understanding clearer – Hematite (ore of iron) has hardness value 5.5-6.5 on Mohs Scale compared to Diamond’s value of 10, but on the scale of toughness Hematite is tougher than Diamond.
Thus, it can be safely said that Hardness and Toughness of a substance are independent of each other.
Is it possible to break a Diamond?
Diamond is extremely hard but is brittle as well, which means it can be broken by single blow. So, special care is required while polishing it.
Earlier, diamonds were polished only partially by rubbing it against another rough diamond. This process of manually polishing diamond is called "bruiting". However, these days laser and other advanced method for polishing have been developed.
Diamond cutting process - also called called Lapidary - is a long and complicated task. It comprises of cutting, shaping and polishing of diamond - resulting into a dazzling diamond that we usually relate to.
Cutting requires detailed study of stone
The hardness of diamond and its ability to cleave depends on the crystallographic direction (on the strength of the bonds between atoms in crystal). The crystallographic structure of the diamond to be polished is analyzed using x-ray diffraction. Such care is taken in order to choose the optimal cutting direction.
Misconception about Diamond's Hardness
Earlier, it was believed that due to its hardness, diamond will not break even if it is hit by hammer. Therefore, it was often hit by hammer to test its hardness and genuineness - that's it's said "half knowledge is very dangerous".
This absurd practice resulted in loss of many precious pieces. But, Thank God this practice was not tried on other famous diamonds like, The Koh-I-Noor or The Blue Hope!
Having said that, now since we know the difference between these two properties, we can say it was not the hardness of diamond that was tested, rather it was its toughness that was tested.
Can Diamond burn or melt?
This is another area of confusion among people that whether diamond can be burnt and/or melted. Well the answer is 'Yes'.
Diamond can be burnt to ashes if subjected to around 700 degree centigrade under normal atmospheric condition (i.e. in presence of oxygen at normal atmospheric pressure). On the other hand, it melt in absence of oxygen and a temperature of around 3500 degree centigrade and a pressure more than million times normal atmospheric pressure.
Diversified Uses of Diamond due to its Hardness
 |
Diamond has transcended across various cultures for its gemological and industrial use. You may like to read about diamond history.
Due to its hardness, diamond is best suited in jewelry as compared to other gems and birthstones. Thus it’s a preferred stone in wedding and engagement rings as it can survive wear and tear of daily use.
But, not all diamonds are used in jewelry. Only 20% of the diamonds mined are of gem quality and remaining are categorized as Industrial grade diamonds. Industrial grade diamonds are used in cutting tools for manufacturing of metals. They are commonly used in diamond-tipped drill bits and saws.
So I am sure, now you would have understood the concept behind hardness of diamond and how it is different from its misunderstood toughness.
Return from Hardness of Diamond to Diamond Information page
Return from Hardness of Diamond to Diamond Jewelry Homepage
I hope you'll not mind sharing this on Twitter, Facebook and with everyone else :)
Feel free to share if something is in your mind and want it to be covered on this site.
My Newsletter
Did you liked this article? Sign-up my FREE weekly newsletter and I'll send you more awesome new additions on this website along with latest jewelry happenings around the world, and download my Jewelry Design Album for FREE!
 |
|